- Your cart is empty
- Continue Shopping
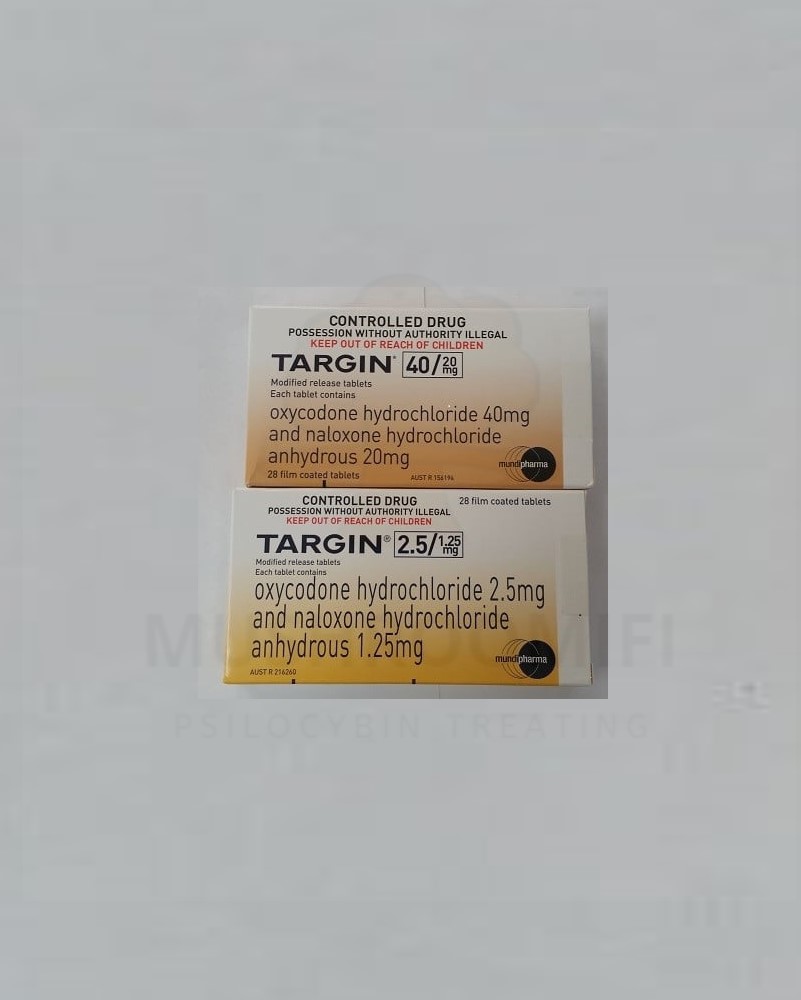
TARGIN X 28 TABLETS
$200.00 – $2,300.00
Quantity, Dosage (MG)
Choose an option / Choose an option
Add to cart
Buy Now
Product Description
Buy TARGIN X 28 TABLETS Online | What it is used for | MushroomiFi.
TARGIN modified release tablet is indicated for: 1. The management of severe pain where: other treatment options have failed, are contraindicated, not tolerated or are otherwise inappropriate to provide sufficient management of pain, and the pain is opioid-responsive; and requires daily, continuous, long term treatment. TARGIN modified release tablet is not indicated for use in chronic non-cancer pain other than in exceptional circumstances. TARGIN modified release tablet is not indicated as an as-needed (PRN) analgesia. The naloxone component in a fixed combination with oxycodone is indicated for the therapy and/or prophylaxis of opioid-induced constipation. ,2. Second line symptomatic treatment of patients with severe to very severe idiopathic restless legs syndrome after failure of dopaminergic therapy. Buy Targin oxycodone online in Australia
2. Scientific discussion
2.1. Introduction
Oxynal 10mg/5mg, 20mg/10mg prolonged-release tablets and Targin 5mg/2.5mg, 10mg/5mg,
20mg/10mg, 40mg/20mg prolonged release tablets and associated names (OXN PR) are fixed
combination products of oxycodone hydrochloride and naloxone hydrochloride. Oxycodone is a semisynthetic opioid which is a narcotic analgesic generally indicated for relief of moderate to severe pain. Buy Targin oxycodone online in Australia
Naloxone is a pure opioid antagonist which is generally used to counter the effects of opioid overdose.
In this case, it added to counteract opioid-induced constipation by blocking the action of oxycodone at
opioid receptors locally in the gut. Buy Targin oxycodone online in Australia
OXN PR is currently indicated in “severe pain, which can be adequately managed only with opioid
analgesics”. Buy Targin oxycodone online in Australia
In the variation procedure the MAH applied for OXN PR to be used in “symptomatic treatment of
patients with moderate to severe idiopathic restless legs syndrome insufficiently treated with
dopaminergic therapy”. During the CMD(h) referral procedure, the proposed restless legs syndrome
indication was restricted to “second line symptomatic treatment of patients with severe to very severe
idiopathic restless legs syndrome after failure of dopaminergic therapy”.
Following the CMD(h) 60 day procedure, the Netherlands was of the opinion that the benefit risk
balance of OXN PR for the restricted indication is still considered negative. The available clinical data,
the proposed adapted product information and the proposed risk minimisation measure are insufficient
to assure that the risks of iatrogenic drug dependence and drug prescription abuse outweigh the. Buy Targin oxycodone online in Australia
Additional Information
| Quantity | 1 Box, 2 Box, 3 Box, 4 Box, 5 Box |
|---|---|
| Dosage (MG) | 2.5/1.25mg, 10/5mg, 30/15mg, 40/20mg |









Reviews
There are no reviews yet.